
As it stands now, there is 1 Na on the left but 2 Na's on the right. In order for this equation to be balanced, there must be an equal amount of Na on the left hand side as on the right hand side. Hydrogen and oxygen are usually balanced last. 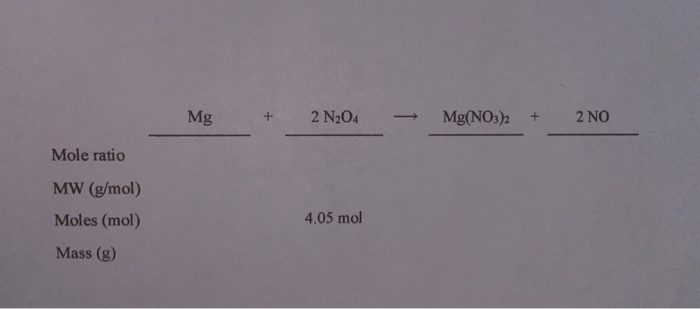
Generally, it is best to balance the most complicated molecule first. Simple chemical equations can be balanced by inspection, that is, by trial and error. Using Trial and Error/Inspection Example #1 (Simple) By changing the scalar number for each molecular formula, the equation may be balanced. In case of net ionic reactions, the same charge must be present on both sides of the unbalanced equation. Thus, each side of the equation must represent the same quantity of any particular element. In a chemical reaction, the quantity of each element does not change.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
